A new clinical trial just announced for 2015 aims to test a “huntingtin lowering” therapy, called an antisense oligonucleotide (ASO), that attacks mutant huntingtin directly. We’re extremely excited—it’s the first-ever human HD trial to fight HD at the root of the problem, and has shown great promise in animal models. What’s the scoop?
A potential drug targeting the HD gene
The announced clinical trial represents a collaboration between California-based Isis Pharmaceuticals, and the Swiss pharmaceutical giant Roche. The drug, called ASO-HTT-Rx, is a therapy that aims to treat HD by targeting the gene itself.
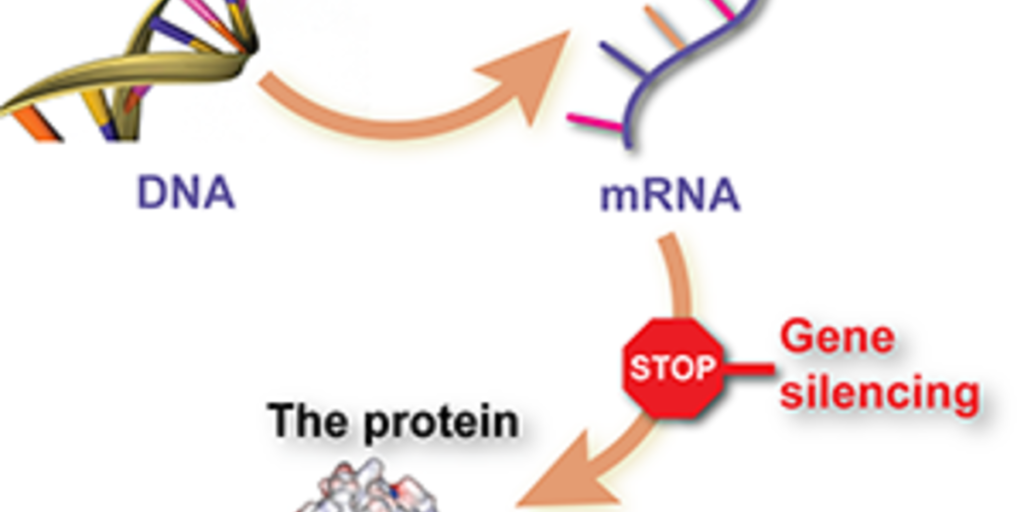
The core of the problem in HD lies in a faulty stretch of DNA—an extra long stretch of CAG building blocks within the huntingtin gene. The instructions contained in the mutant gene are first copied into an intermediate ‘messenger’ copy, from which the harmful protein is made. So, the mutant gene is the root of the problem, but it’s only bad because cells use the information in the gene to make a harmful protein.
The strategy behind ASO-HTT-Rx is to “shoot the messenger,” attacking the intermediate step between gene and protein by causing the destruction of the messenger copy.
These types of drugs are called ‘antisense oligonucleotides’, or ASOs. They’re a synthetic DNA-like molecule that can enter cells, stick to the mutant huntingtin RNA message, and cause its degradation. If it works as predicted, this therapy will stop huntingtin protein from being made—a “huntingtin lowering” strategy that, in the long term, has the potential to slow or to halt disease progression.
Every ASO has a basic structure that can be tweaked to help it stick to the right target message, ignoring the thousands of other messages in the cell. It’s a bit like when you go to the hardware store to have a key copied—the clerk selects the matching blank key and then etches the correct sequence of grooves to fit your lock, but none of your neighbors' locks. In the case of ASOs, scientists at Isis use one of their established ‘backbone’ molecules and customize it, causing the drug to attack only the Huntingtin message.
This all sounds like cutting-edge technology, but the great news is that Isis has tested ASO-based drugs in human brains before, in experimental treatments for ALS (Lou Gehrig’s, or motor neuron, disease) and SMA (spinal muscular atrophy), and there have been no reported issues with safety.
Drug delivery
One major way that ASOs differ from conventional drug treatments is that they cannot be taken orally as a pill, but must be delivered directly to the nervous system.
To get into the brain, ASO-HTT-Rx will be administered via a needle inserted into the fluid-filled space below the lower spinal cord. If that sounds extreme, rest assured that this method is routinely used in many areas of medicine. A similar strategy is used by cancer doctors to deliver chemotherapy drugs to patients with brain tumors. More commonly still, many women receive similar ‘epidural’ delivery of pain drugs during child birth.
A major advantage of drugs like ASO-HTT-Rx is that researchers believe it can be administered intermittently and still be effective. Scientists have dubbed this approach to intermittent treatment in HD a ‘huntingtin holiday’; aiming to give the brain a healing break from the damage caused by the mutant Huntingtin protein.
Once injected into the spinal fluid, it takes about 4 to 6 weeks for ASO-HTT-Rx to have its effect, and from animal studies we think that the silencing will then last for about 4 months. At the moment, the study is designed to have patients receive the drug treatment once a month.
Safety first
The decision to push forward with a clinical trial starting in 2015 is the result of more than ten years of work involving many researchers in both academia and industry. When treated with ASO-Htt-Rx, lab mice that model HD show robust benefits—even treating them with ASOs after they show symptoms leads to improvements in their brains and behavior.
Isis researchers and Prof Sarah Tabrizi, University College London and global head of the ASO-HTT-Rx study, emphasize this first clinical trial is designed purely to evaluate its safety.
This is a critical point. As exciting as this science is, the first planned trial is strictly designed to understand whether the drug is safe. Directly injecting any drug into the nervous system cannot be done lightly—all the potential risks and side effects have to be exhaustively examined in a small number of volunteer patients.
Anatomy of a clinical trial
All Phase I clinical trials aim first and foremost to evaluate safety and tolerability of new drugs. This means that a small group of patients (probably around 36) will be treated with differing amounts of ASO-Htt-Rx, in order to determine whether the drug has adverse side effects, and to help find the optimal dose.
About 25% of the patients involved will receive a placebo injection (one that does not contain any drug) to serve as a comparison group. Doctors will be monitoring the patients’ symptoms in response to the treatment, but the main focus of this trial is about whether the new drug is safe.
Once a drug has been deemed safe in Phase I, it can progress to Phase II. It’s at this second step that physicians can recruit larger numbers of patients and begin to examine how effective the drug is in treating disease symptoms. Having established safety and dose in the first trial, researchers can now ask the question, ‘does this treatment make people’s HD symptoms better (or worse!)’?
If the results of the Phase II trial are positive, a Phase III trial involves even greater numbers of patients, carefully examining the drug’s side effects, effectiveness, and safety. A successful Phase III trial is the kind of result that drug companies need to get a drug approved by national regulatory agencies.
What does this mean for HD patients?
The entire process of bringing a drug to market takes years, even if everything goes perfectly. If ASO-Htt-Rx turns out to be safe in the just-announced Phase I study, this first result is just the beginning of a long road in the clinic. Each trial along that path, Phases I, II and III, will involve a relatively small number of treated patients. Furthermore, within each of these trials, some of the volunteers will receive placebo treatment to serve as a comparison for patients receiving HTT-ASO-Rx.
Patients in the Phase I trial of HTT-ASO-Rx will be recruited from just a few medical centers in Europe and Canada. A small set of designated HD research centers, to be announced in early 2015, will seek to recruit trial participants. The recruits will be people in the early stages of HD, but the specific enrollment criteria, locations and timing are not yet public information. We know for sure that a lot of detailed planning is happening behind the scenes, and that everyone is working really hard to advance this trial as quickly as possible.
Safety, uncertainty, hope
In summary, we’re excited that the first human trial of a drug that targets the root cause of HD will start as early as next year. This excitement is tempered by the realization that this first trial is all about safety, involves only a very small number of HD patients, and that many details of the study are still uncertain.
Drugs similar to ASO-Htt-Rx have safely restored healthy behaviors in animal models of HD, and moving the treatment into humans is an exciting step for the whole HD community. We’ll be following the trial’s progress with a keen eye, so that hopefully we can provide intermittent doses of cautious optimism.